UK Will Infect Dozens Of Healthy Volunteers In World's First COVID-19 'Challenge Trial'
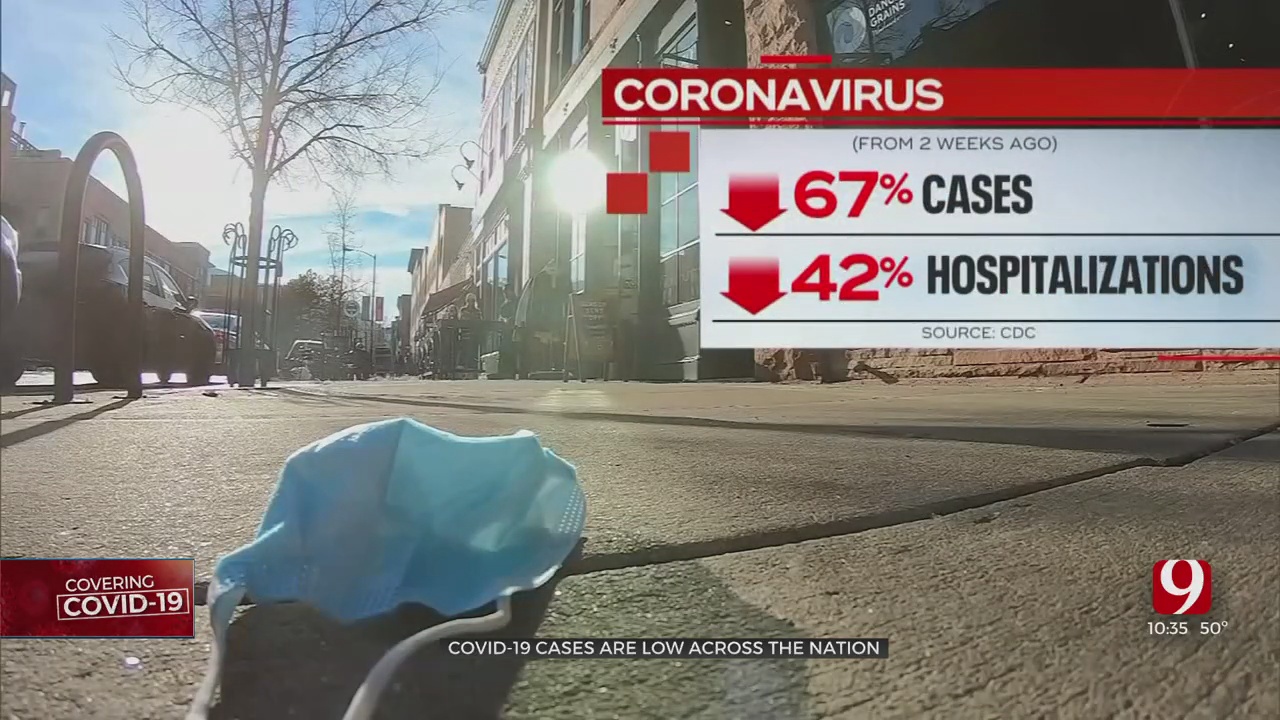
England is set to become the first country in the world to intentionally infect healthy volunteers with COVID-19 in what's known as a "human challenge study." The country's medical ethics body approved the trial Wednesday.Thursday, February 18th 2021, 12:56 pm
England is set to become the first country in the world to intentionally infect healthy volunteers with COVID-19 in what's known as a "human challenge study." The country's medical ethics body approved the trial Wednesday.
Funded by the British government, the $47 million study will play a crucial role in continuing the development of effective coronavirus vaccines and treatments, officials said.
The first-of-its-kind challenge study is expected to begin within the next month, involving up to 90 carefully selected, healthy volunteers aged 18-30. They will be compensated for their time.
During the trial, the volunteers will be exposed to the smallest amount of the virus needed to cause infection, in a "safe and controlled environment," to further understand its effects. The study will use the version of the virus that's been circulating in the U.K. since March 2020, not the new variants.
Officials noted that medics and scientists will closely monitor the participants 24 hours a day, and emphasized that the March 2020 strain "has been shown to be of low risk in young healthy adults."
They hope to identify the smallest amount of virus needed to cause infection. The trial will also help doctors and scientists understand how the immune system reacts to the virus and identify factors that affect how it is transmitted.
"While there has been very positive progress in vaccine development, we want to find the best and most effective vaccines for use over the longer term," Business Secretary Kwasi Kwarteng said in a statement. "These human challenge studies will take place here in the U.K. and will help accelerate scientists' knowledge of how coronavirus affects people and could eventually further the rapid development of vaccines."
The controversial study is different from typical vaccine trials, in which participants are given a vaccine or placebo and then told to go about their everyday lives. In that case, researchers expect that some volunteers will eventually be exposed to the virus naturally.
Challenge trials, however, guarantee exposure in the hopes of speeding up results. They have been used in the past to study diseases like malaria, typhoid, cholera, norovirus and influenza.
After the first trial, a small number of volunteers could be given vaccine candidates that have passed clinical trials, to identify which are most effective. This week, the U.K. hit its target of vaccinating 15 million people with their first dose.
"We have secured a number of safe and effective vaccines for the UK, but it is essential that we continue to develop new vaccines and treatments for COVID-19," Clive Dix, interim chair of the Vaccines Taskforce, told BBC News. "We expect these studies to offer unique insights into how the virus works and help us understand which promising vaccines offer the best chance of preventing the infection."
More Like This
February 18th, 2021
March 7th, 2022
March 7th, 2022
February 19th, 2022
Top Headlines
April 26th, 2024
April 26th, 2024
April 26th, 2024