Pfizer Submits Initial Data To FDA For COVID-19 Vaccine In Children 5 To 11
Last week, Pfizer came out with results from their phase 2 and 3 trial of more than 2,000 children from ages 5 to 11.Tuesday, September 28th 2021, 4:13 pm
Last week, Pfizer came out with results from their phase 2 and 3 trial of more than 2,000 children from ages 5 to 11.
Pfizer said that the data showed similar results in efficacy for those 16 to 25. The dosage for the 5 to 11 is just a smaller dose.
The data results have been shared with the FDA for the agency’s initial review.
Pfizer is expected to submit a formal request for emergency use authorization (EUA) for its COVID-19 vaccine in children 5 to under 12 years of age after releasing data from its trials.
Submissions to the European Medicines Agency (EMA) and other regulatory authorities are also planned.
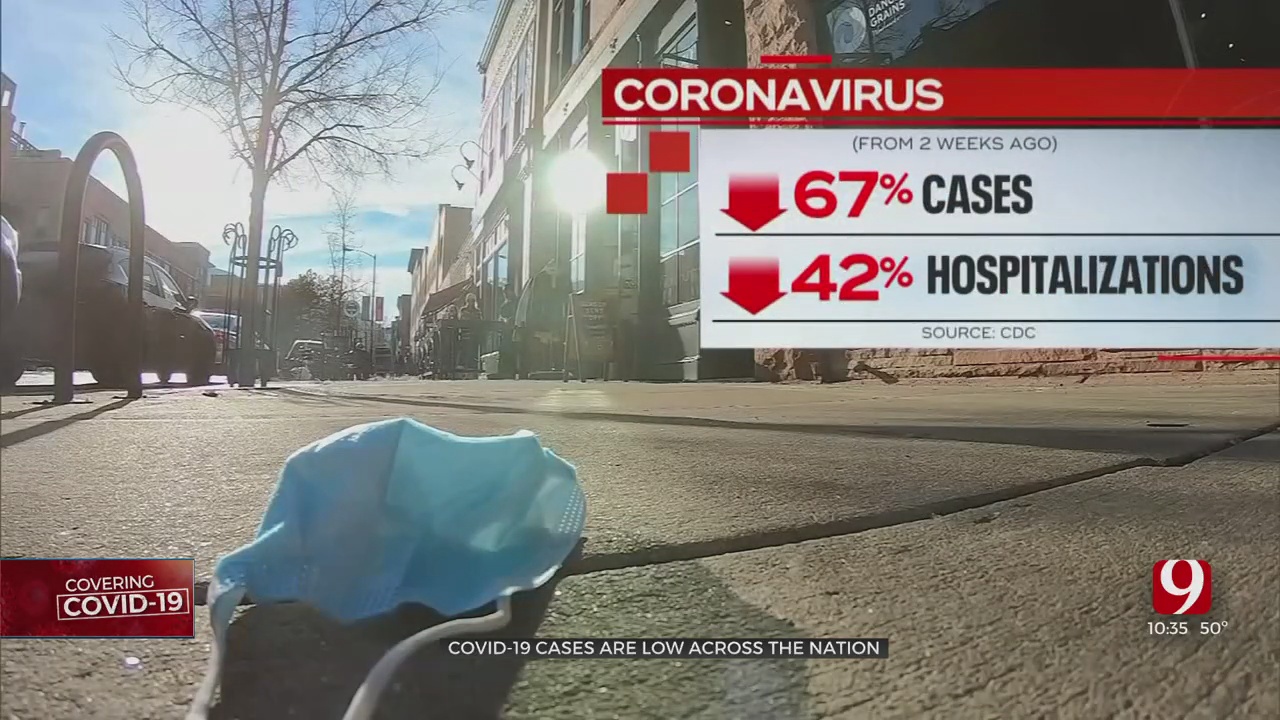
Pediatricians are hopeful for the potential to be able to vaccinate kids ages 5 to 11, as kids under 12 made up just under 10% of cases in the state last week.
"Our hospital system is strained in pediatrics, too, and studies have shown unvaccinated teenagers have a 10x more likely to be hospitalized than vaccinated teenagers," said Dr. Ashley Weedn, a pediatrician with OU Health.
Weedn said the delta variant is affecting children differently as they have seen higher numbers of children end up in the hospital as compared to the beginning of the pandemic.
Right now, only kids 12 and older are able to get the Pfizer vaccine, but Weedn said she is hopeful for those 5 to 11 to be able to roll up their sleeves soon.
"The smallest dose of 10 micrograms was selected as the preferred dose that mounted a sufficient antibody response with minimal side effects," said Weedn.
The data will need to be analyzed by the FDA, and the CDC will need to make their final recommendations before the green light is given.
"As a mom and not just a pediatrician, but as a mom of schools age kids, I am excited for this," said Weedn.
Right now, there is no exact date when the decision will come but Weedn is hopeful it will be soon.
"What I am personally looking at is hoping for Halloween, but if we have it before Thanksgiving or the holidays, I'll be thrilled," said Weedn.
More Like This
September 28th, 2021
March 7th, 2022
March 7th, 2022
February 19th, 2022
Top Headlines
June 1st, 2025
June 1st, 2025