Day 6: Johnson & Johnson Goes On The Offensive In State's Opiod Trial
The defense went on offense Tuesday. Day 6 of the state’s opioid trial saw defendant Johnson & Johnson paint a picture of its drug marketing actions that stands in stark contrast to the one portrayed so far by the state.Wednesday, June 5th 2019, 5:07 am
The defense went on offense Tuesday. Day 6 of the state’s opioid trial saw defendant Johnson & Johnson paint a picture of its drug marketing actions that stands in stark contrast to the one portrayed so far by the state.
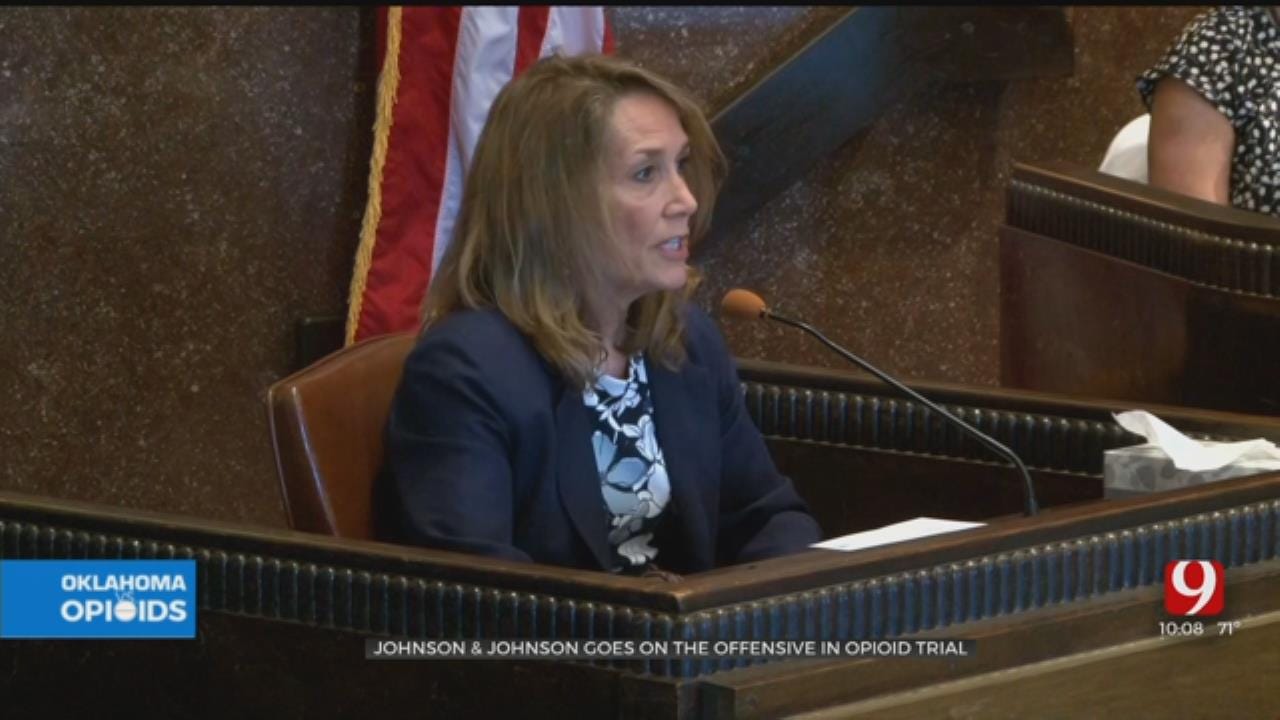
On the witness stand for a fifth consecutive day was Kimberly Deem-Eshelman, who is serving as Johnson & Johnson’s corporate representative during the trial, but who actually is a longtime employee of Janssen Pharmaceuticals, a Johnson & Johnson subsidiary. Under questioning from Oklahoma City attorney Larry Ottaway, Deem-Eshelman provided testimony aimed at showing the drugmaker brought its opioid pain relievers to the Oklahoma market responsibly and through proper channels.
“Can Janssen directly supply opioid products to residents of Oklahoma?” Mr. Ottaway asked.
“No, sir,” replied Deem-Eshelman.
Mr. Ottaway spent considerable time Tuesday morning showing that Johnson & Johnson took very seriously its responsibility to properly inform doctors and patients about any risks associated with their drugs.
“What is the first thing the label says?” queried Ottaway.
“Warning - may be habit forming,” responded Deem-Eshelman.
The defense team provided evidence to show that, not just on product labels, but also promotional materials, the company provided exhaustive FDA-approved warnings for its opioid products.
“Duragesic exposes users to risk of addiction, abuse, and misuse, which can lead to overdose and death,” read Deem Eshelman from a Duragesic label, one of many entered into evidence.
Janssen launched Duragesic, a fentanyl patch, in 1991. In the late 1990s, the company re-launched it for treatment on non-cancer pain; it went off patent in 2005. Nucynta, generically known as tapendadol, was rolled out in 2009. An extended release version, Nucynta ER, came out two years later.
In testimony Tuesday, the company explained that it formulated Nucynta in a way to try and minimize abuse.
“We needed to bring a drug to market in a responsible way,” said Deem-Eshelman, “so the extended release formulation was a significant development to bring that out in a capsule that was very difficult to abuse, difficult to crush, difficult to manipulate.”
Deem-Eshelman also testified at length about Jannsen and its corporate parent’s multi-faceted efforts to educate doctors about their drugs, through speakers bureaus, continuing medical education and through its drug reps.
“Our goal is to educate the provider on the benefits and the risks associated with our medications,” Deem-Eshelman told Judge Thad Balkman. “Ultimately, it is the physician’s decision what medicine they use for their patient.”
Following the adjournment of court, Oklahoma Attorney General Mike Hunter addressed the media and said the defendant’s guilt or innocence can best be judged by its actions, not its words.
Hunter says, at the height of the opioid crisis, between 2009 and 2011, Jannsen sales reps’ visits to Oklahoma doctors shot way up.
“Under the corporate euphemism of ‘education,’ [the company] clearly influenced, targeted, hooked, and leveraged doctors into buying their opioids,” Hunter stated.
Defense attorneys say they still have about 90 minutes of cross-examination for Deem-Eshelman, after which that state will be able to question her again on re-direct.
The trial is scheduled to resume at 9:00 a.m Wednesday.
More Like This
March 22nd, 2024
March 14th, 2024
February 9th, 2024
Top Headlines
April 26th, 2024
April 26th, 2024
April 26th, 2024