Local Health Expert Talks About Pfizer Vaccine After FDA Approves Expansion Of Eligibility To Kids Ages 12 To 15
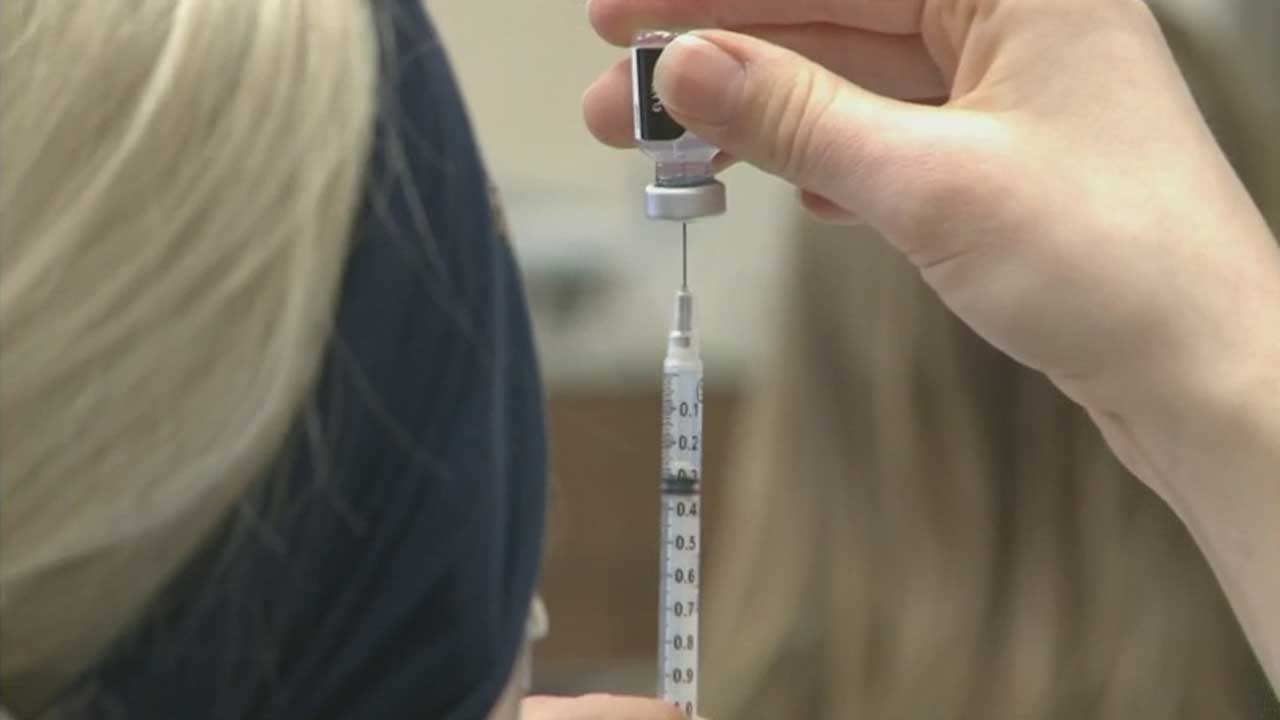
The Food and Drug Administration has granted emergency use authorization for the Pfizer COVID-19 vaccine to allow children ages 12 to 15 to be eligible.Tuesday, May 11th 2021, 10:04 am
The Food and Drug Administration has granted emergency use authorization for the Pfizer COVID-19 vaccine to allow children ages 12 to 15 to be eligible.
The company said the vaccine has 100% efficacy rates shown in this new age group of 12 to 15.
"It a safe vaccine, and it's effective. I don't see a lot of downsides," said Carlos Blanco, the president and CEO of the Lynn Health Science Institute.
His team has been studying COVID-19 vaccines since the beginning.
By looking at the data from the Pfizer vaccine, he said this is a safe vaccine for children to use.
This new age group would make 17 million young people eligible for the vaccine.
In a study done by the Kaiser Family Foundation, only 3 in 10 parents with children in this age group are ready to give their children the vaccine.
In the Pfizer vaccine trials with 2,200 volunteers, there were zero cases of COVID-19 in the children who got the vaccine compared to 18 cases among those who got the placebo.
As for side effects, those are showing to be the same in younger populations across the board, with the second dose being what might cause fatigue, fever, and sore arm.
"I'm a parent. Would I be worried about vaccinating my child? Not in the slightest," said Blanco.
The Lynn Institute plans to run Moderna trials on a younger population of 2- to 12-year-olds in just a few weeks.
The state and the Oklahoma City-County Health Department are awaiting for recommendations from the CDC's ACIP before administering the vaccine to this age group.
More Like This
May 11th, 2021
March 8th, 2022
January 21st, 2022
January 13th, 2022
Top Headlines
April 24th, 2024
April 24th, 2024
April 24th, 2024