Local Doctor Explains Blood Clots Caused By Johnson & Johnson Vaccine
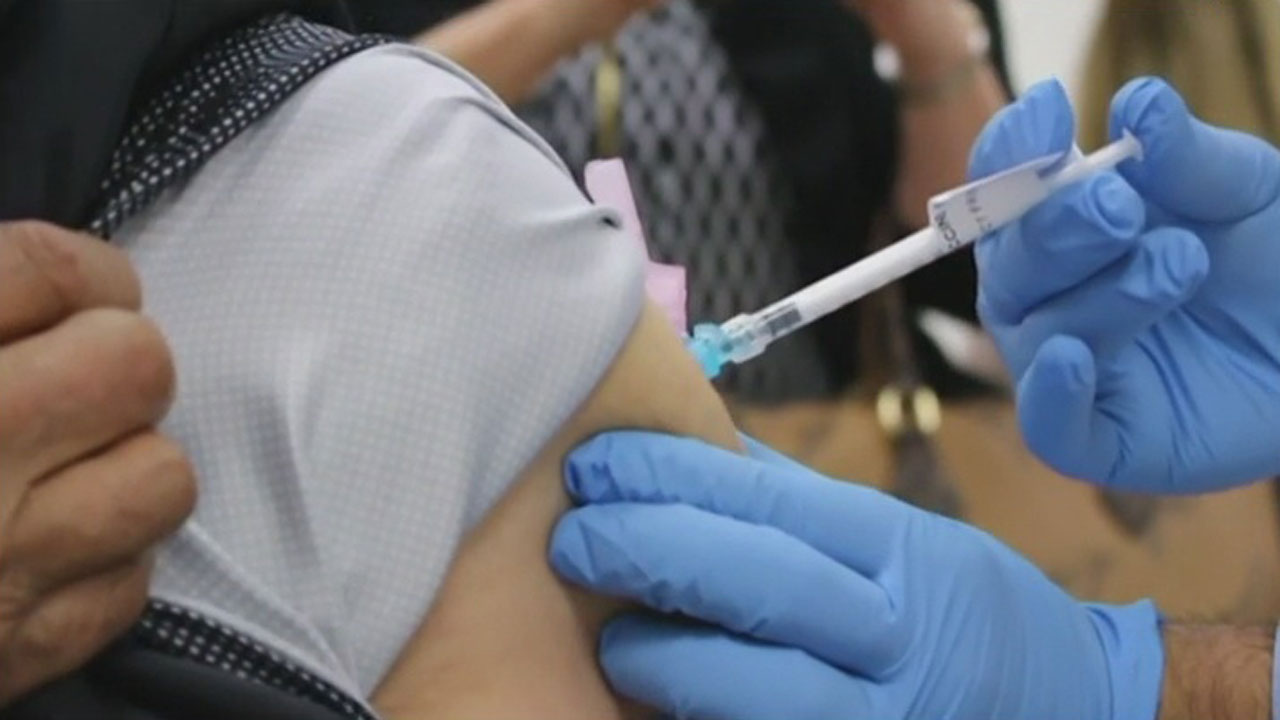
This week the FDA & CDC paused Johnson & Johnson covid-19 vaccine distribution in the U.S. due to a rare but serious blood clotting condition.Friday, April 16th 2021, 5:24 am
OKLAHOMA CITY -
This week the FDA & CDC paused Johnson & Johnson covid-19 vaccine distribution in the U.S. due to a rare but serious blood clotting condition.
OU's chief COVID-19 officer, Dr. Dale Bratzler says the Johnson & Johnson vaccine is similar to the AstraZeneca vaccine. They are both what is called a viral vector vaccine. This means they both use a non-replicating virus that injects a small piece of DNA from the covid virus that causes your body to respond and make spiked protein. Your body then creates antibodies to fight against the virus in the future.
Pfizer and Moderna are manufactured differently. They are what is known as an mRNA vaccine, a new type of vaccine.
According to Dr. Bratzler, “We have a pretty good sense of what may be happening. The question is why is it happening.”
Dr. Bratzler says a possible cause of clotting with the J&J vaccine may be something associated with the vaccine, or the immune response stimulated by the vaccine affects or attaches to a platelet factor four.
“There are some very good articles from researchers particularly about the AstraZeneca vaccine that show that there is some aspect of the vaccination, whether it is the viral vector, whether it's the DNA within in the virus, whether it is the immune response that you get from the vaccine. ”
He says that something about the vaccine activates your platelets.
“Platelets are small calls that circulate in your blood all of the time. They are responsible for blood clotting, and they typically don't stick together and don’t clot but “something about getting these vaccines seems to activate those platelets, they come together they clump they clot, and it creates those clots that we have seen in the AstraZeneca and Johnson and Johnson vaccine,” says Dr. Bratzler.
He also says the issue is very rare.
“Less than one person out of a million doses of vaccine have had this reaction in the United States, but I completely understand why the FDA is taking the time to pause," he says.
The Johnson and Johnson suspension is expected to last until at least next week when a CDC advisory committee has its next meeting on the investigation.
More Like This
March 8th, 2022
January 21st, 2022
January 13th, 2022
Top Headlines
April 24th, 2024
April 24th, 2024